Histology is the study of cell structure and tissues by examining a thin section under the microscope. The tissue sections are carefully prepared using special processes called histological techniques. For light microscopy, paraffin and frozen sections are the most commonly used techniques.
With the paraffin technique the tissue is fixed, dehydrated, cleared and embedded in paraffin wax and sectioned using a microtome (Paraffin section thickness is about 5µm). With frozen sections, fresh or fixed tissue is frozen in liquid nitrogen or dry ice, and then sectioned with a cryostat (frozen section thickness is approximately 15-30µm).
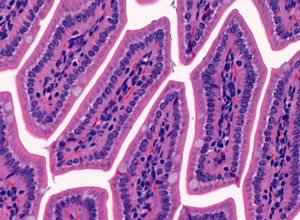
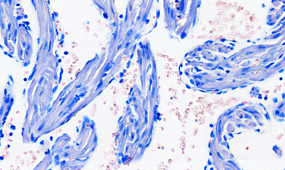
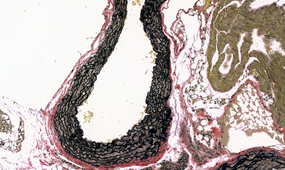
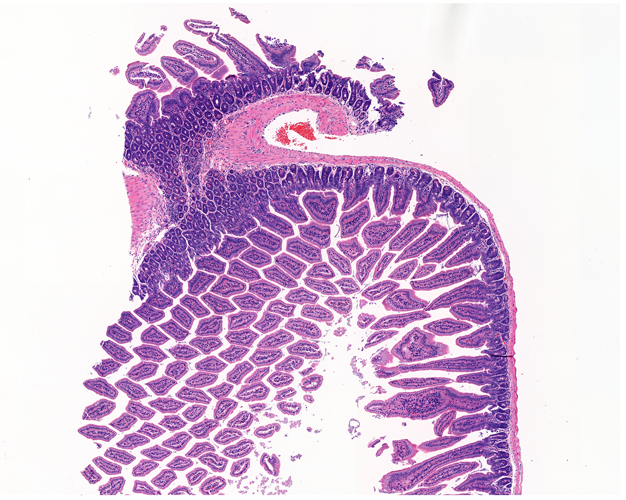
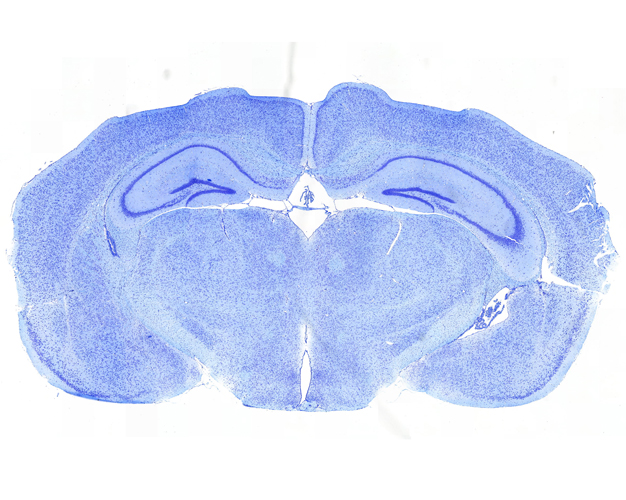
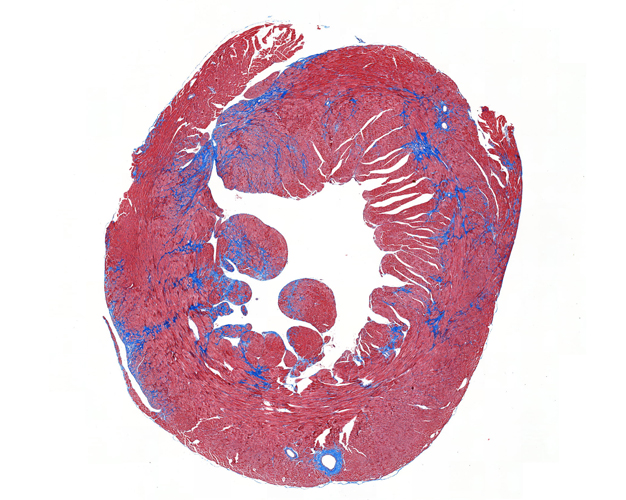
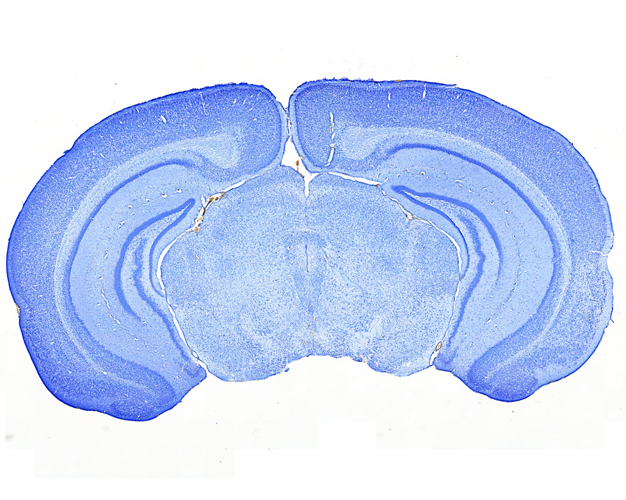
Once the sections are prepared using one of these techniques, they are stained to provide color and contrast to the cell structure and tissue. Staining usually works by applying a dye which stains some of the cell components with a bright color, together with another dye that stains the rest of the cell in a different color.
Our goal is to prepare from your fixed tissue paraffin or frozen sections and mount them on slides ready for immunostaining. We also offer routine histochemical staining with Hematoxylin-Eosin, Nissl, Masson Trichrome, Oil Red, Sirius Red, Perl’s Prussian Blue.
Our Histology Service Laboratory also works with the researcher to develop new histological approaches to demonstrate changes in cell/tissue morphology.
Services provided:
| ▪ | Tissue processing: preparation of paraffin-embedded samples. |
| ▪ | Sectioning: serial and step sectioning of paraffin-embedded samples and frozen samples by microtome or cryostat respectively. |
| ▪ | Staining: H&E, Masson trichrome, Nissl, Oil Red, Sirius Red, Perl’s Prussian Blue, Giemsa, Van Gieson. |
| ▪ | Assistance with histological protocol optimization. |
| ▪ | Development of histological staining. |
In addition, we can provide:
| ▪ | Digitization of the whole slide using our automated whole slide scanner. |
| ▪ | Quantitative image analysis. |
Histology services: How to start?
Please follow the instructions in this file. In case of questions please contact: Katren Sakran tel. +972-73-378-1109.
For any question regarding data acquisition or image analysis please contact Maya Holdenberger.